Aerogel insulation for buildings
Contents |
[edit] Introduction
Aerogels are synthetic low-density materials with unique physical properties. They are formed by removing the liquid from a gel under special drying conditions, bypassing the shrinkage and cracking experienced during ambient evaporation. This creates a solid three-dimensional nanoporous structure containing 80-99% air.
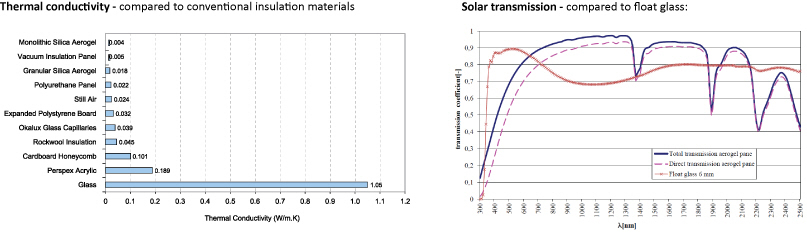
Due to their high porosity, aerogels exhibit the lowest thermal conductivity of any solid, whilst being transparent to light and solar radiation. Aerogels are often cited as a promising material for translucent insulation applications. They can made from practically any material, although the most common form is silica aerogel which can be produced as granules or in solid (monolithic) tiles.
Commercial products for the building sector include:
- Cavity insulation.
- Glazing units and cladding systems containing granular aerogel.
- Translucent and opaque insulation boards, blankets and tensile roof membranes embedded with aerogel particles.
Transparent monolithic silica aerogel has been cited as the ‘holy grail’ of future glazing technology, with the potential to achieve U-values as low as 0.1 W/m2.K. However, research and development into monolithic glazing is limited due to the high cost of production, long processing time and the difficulty of creating large uniform samples with complete transparency.
[edit] How it works
The total thermal conductivity of porous insulation depends on the heat transfer through convection in the pores, conduction through the solid and pores, and radiation. Typically, pores within conventional insulation are over 1mm wide, allowing gas molecules to move freely and transfer thermal energy by convection. By comparison, pores within aerogel can be as small as 20-40nm (even smaller than the ‘mean free path’ of air at 60-100nm). As a result, individual air molecules within the pores have no space to transfer thermal energy by convection.
Conduction through the solid structure and air molecules within aerogel is also minimal. With little space for convection, air molecules constantly collide with the walls of the pores, suppressing gas conduction. Furthermore, as aerogel only contains 0.1-5% silica and the thermal conductivity of air is very low, heat transfer is minimal and conduction in the gas will diminish with any decreases in pressure. A vacuum inside the pores results in the best insulating properties, with thermal conductivity of 0.004 W/m.K (ten times better than conventional insulation).
The amount of radiative heat transfer through aerogel is dependant on the intensity and wavelength of the thermal radiation, the optical properties of the material, the size and shape of its pores and its overall thickness. At ambient temperature, the nanosized pores and particles provide effective attenuation of infrared thermal radiation due to high levels of absorption and reflection.
The optical and infrared properties of silica aerogels have been well studied. Silica aerogels can be considered transparent insulation materials that effectively transmit solar light, but block thermal infrared radiation.
The material exhibits high translucency, often accompanied by a slight bluish haze. This can be attributed to ‘Rayleigh scattering’, an optical phenomenon that occurs when light scatters off particles smaller than the wavelength of light, where shorter wavelengths in the blue spectrum are most easily scattered.
[edit] Environmental impact
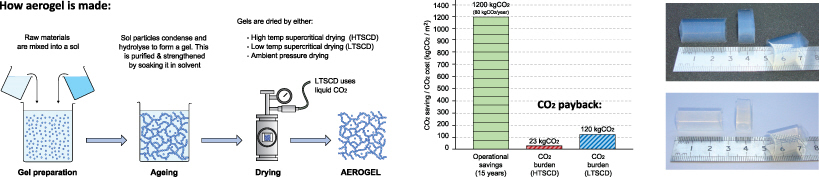
To make aerogel involves three key steps: gel preparation, ageing and drying. The aim is to create a gel, strengthen and purify it, then remove all liquid from the pores without collapsing the solid structure (achieved through super-critical or sub-critical drying techniques). These processes typically involve mixing reasonably toxic chemicals and undertaking complex diffusion controlled processes that consume a lot of solvent. Furthermore, the final step is often accompanied by intensive drying processes which may consume large amounts of energy and CO2.
Despite this, In 2008, the two major manufactures of silica aerogel both received ‘Silver’ cradle-to-cradle environmental awards from McDonough Braungart Design Chemistry (MBDC) for their aerogel production. MBDC claim to evaluate a products complete formulation, energy use, water use and recycling potential when assessing environmental impacts. However, the data from these studies is confidential, making it difficult to assess the rigour and validity of the results.
To investigate these issues further, Mark Dowson conducted a first hand life cycle assessment (LCA) of silica aerogel following ISO standards. Working in collaboration with the University of Bath (who manufacture aerogel as part of an optics research programme) Mark made some aerogel himself, and measured the raw material and electricity use across the production line.
The findings from the study demonstrated how the energy and CO2 required to manufacture high performance silica aerogel insulation can be recovered within 0-2 years (when comparing results to the in-use savings arising from retrofitting to single glazing). Key factors influencing the environmental payback were the efficiency of production and re-use / recycling of materials such as liquid CO2 and solvents.
[edit] Potential developments
Within Buro Happold, Mark Dowson undertook an Engineering Doctorate (EngD), in collaboration with Brunel University, focusing on developing and testing new building fabric technologies incorporating translucent granular aerogel encapsulated inside clear polycarbonate sheets to improve the thermal performance of new and existing buildings. Three technologies have been developed, with promising initial results:
- Retrofitting polycarbonate panels filled with aerogel granules to existing windows to improve their thermal performance.
- Retrofitting a translucent aerogel panel to the outside of a concrete wall to trap solar energy that can be used to passively warm a building.
- An aerogel solar collector incorporating a translucent polycarbonate cover filled with granular aerogel insulation.
See Transparent insulation materials for more information.
This article is based on a paper written by Mark Dowson of --Buro Happold. An online version of Mark's EngD thesis can be downloaded at the Brunel University website: http://bura.brunel.ac.uk/bitstream/2438/7075/3/FulltextThesis.pdf
[edit] Related articles on Designing Buildings
- Advanced phase change materials.
- Advanced phase change materials industry.
- Aerogel market.
- BREEAM Insulation.
- Insulation specification.
- Life cycle assessment.
- Phase change materials.
- Sarking.
- The thermal behaviour of spaces enclosed by fabric membranes.
- Thermal comfort.
- Transparent insulation materials.
- Types of insulation.
- U-values.
[edit] External references
- Mark Dowson, David Harrison, Salmaan Craig, Zachary Gill. International Journal of Sustainable Engineering, Taylor & Francis Group, March 2, 2011.
- Mark Dowson, Michael Grogan, Tim Birks, David Harrison, Salmaan Craig. Third International Conference on Applied Energy, Perugia, Italy, May 18, 2011
- Mark Dowson, Ian Pegg, David Harrison. 2011 Conference for the Engineering Doctorate in Environmental Technology, June 21, 2011
- http://www.aerogel.org/
- http://en.wikipedia.org/wiki/Aerogel
- Aspen Aerogels: http://www.aerogel.com/
- Proctor: https://www.proctorgroup.com/products/spacetherm
- Kalwall: http://www.kalwall.com/
- https://www.idtechex.com/research/reports/aerogels-2019-2029-technologies-markets-and-players-000644.asp
Featured articles and news
We're expanding our collaborative mission by launching DB Intelligence, an exclusive market research advisory panel. Built environment professionals can now get paid to share their expertise on industry trends, products and services.
Panel members receive direct financial incentives for participating in research projects like short surveys, 1-2-1 interviews and focus groups. Register today to shape the future of the construction sector.
Building Safety Wiki Interviews
Chief executive of the British Woodworking Federation.
Planning condition discharge in England and Wales
A brief exoplanation from a building compliance expert, with further links.
Overheating guidance and tools for building designers
Guidance for dealing with element of building fabric control that have increasing importance.
Shading for housing, a design guide
From the Good Homes Alliance and British Blind and Shutter Association.
UK Standard Skills Classification (SSC)
A shared framework for describing skills needs.
Social media ban consultation comes to close
CIOB urges UK Government to consider social media’s role in careers guidance in ban debate.
The latest of eight Skills England apprenticeship units
The addition of battery manufacturing welcomed by ECA with a warning about the risks of fast-tracked apprenticeship units.
Building Control Independent Panel final report
A precis of a key report led by Dame Hackitt with full recommendations and link to the government response.
Building Safety recap April, 2026
A short and longer run-through of the month, with links to further information and sources.
CIAT May 2026 briefing.
From medieval scribes to modern word art.
ECA welcomes crackdown on late payment and push for clean energy, whilst CIOB seek fixed cladding removal timeframes.
Cyber Security in the Built Environment
Protecting projects, data, and digital assets: A CIOB Academy TIS.
Managing competence in the built environment
ITFG publishes new industry guide on how to meet the ICC principles.























Comments
To start a discussion about this article, click 'Add a comment' above and add your thoughts to this discussion page.