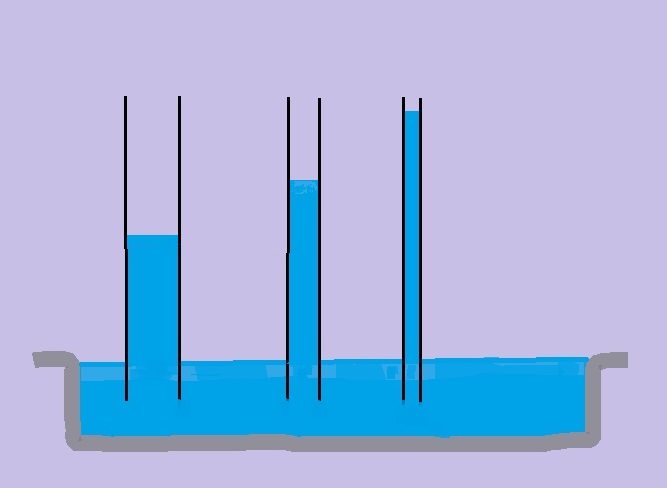
Capillary action
|
| Capillary action causes the water in the thinnest tube to rise to a higher level than in the other tubes |
[edit] Introduction
Capillary action is a phenomenon associated with surface tension, whereby liquids can travel – horizontally or vertically (against the force of gravity) in small spaces within materials. It is sometimes referred to as capillary attraction, capillarity or wicking.
The movement is due to the surface tension that results when liquid or moisture is contained within very fine spaces or tubes (capillaries). Essentially, the liquid is attracted to the sides of the container; the smaller the space, the greater the attraction. Examples of capillarity include the action observed when a paper towel or blotting paper absorb water, and the way oil travels up a wick in oil lamps.
[edit] Rising damp
Rising damp in concrete and masonry is also the result of capillary action. When building materials such as most brick types, some stones, concrete blocks and plaster come into contact with moisture, the water adheres to the pores of the material’s capillaries. If the adhesive force between the water molecules and the material is greater than the cohesive force existing between the water molecules themselves, the water rises up the tube through capillary action.
Typically, damp can rise up to around 1m above its source. It is usually prevented from doing so by the installation of a damp-proof course (DPC), typically a polymerised rubber material such as bitumen polymer. Installed as the brickwork goes up and bedded both sides with mortar, the bond between DPC, mortar and walling material creates a barrier to moisture rising through capillary action.
Capillary action is also seen in many plants and trees.
Leonardo da Vinci is regarded as being the first person to observe and record capillary action.
Technical paper 35: Moisture measurement in the historic environment, published by Historic Environment Scotland in 2021, defines capillarity as: ‘The tendency of a liquid in an absorbent material to move as a result of surface tension.’ It suggest that capillary rise is: ‘...controlled by capillarity - the rise of a liquid in an absorbent material above the level that would be influenced solely by atmospheric pressure.’
[edit] Related articles on Designing Buildings
- Blockwork.
- Building damp-free cavity walls.
- Building science.
- Capillary active material.
- Capillary break.
- Chemical injected DPC.
- Damp.
- Damp proof membrane.
- Defects in brickwork
- Defects in stonework.
- Penetrating damp.
- Rising damp.
- Rising damp in walls - diagnosis and treatment (DG 245).
- Tempering heating.
- Vapour barrier.
Featured articles and news
We're expanding our collaborative mission by launching DB Intelligence, an exclusive market research advisory panel. Built environment professionals can now get paid to share their expertise on industry trends, products and services.
Panel members receive direct financial incentives for participating in research projects like short surveys, 1-2-1 interviews and focus groups. Register today to shape the future of the construction sector.
Planning condition discharge in England and Wales
A brief exoplanation from a building compliance expert, with further links.
Overheating guidance and tools for building designers
Guidance for dealing with element of building fabric control that have increasing importance.
Shading for housing, a design guide
From the Good Homes Alliance and British Blind and Shutter Association.
UK Standard Skills Classification (SSC)
A shared framework for describing skills needs.
Social media ban consultation comes to close
CIOB urges UK Government to consider social media’s role in careers guidance in ban debate.
The latest of eight Skills England apprenticeship units
The addition of battery manufacturing welcomed by ECA with a warning about the risks of fast-tracked apprenticeship units.
Building Control Independent Panel final report
A precis of a key report led by Dame Hackitt with full recommendations and link to the government response.
Building Safety recap April, 2026
A short and longer run-through of the month, with links to further information and sources.
CIAT May 2026 briefing.
From medieval scribes to modern word art.
ECA welcomes crackdown on late payment and push for clean energy, whilst CIOB seek fixed cladding removal timeframes.
Cyber Security in the Built Environment
Protecting projects, data, and digital assets: A CIOB Academy TIS.
Managing competence in the built environment
ITFG publishes new industry guide on how to meet the ICC principles.
The UK's campaign to reduce noise pollution: Mythbusting, articles and topic guides.